|
Make paper plate atom models, we used these to learn about isotopes. Teach Beside Me has an amazing Periodic Table Battleships game. Titanium is very strong and very, very light! Ideas for learning about the Periodic Table Neon is the least reactive element in the Periodic Table and is used in neon signs. Mercury is the only transition metal that is a liquid at room temperature.īismuth is diamagnetic. Helium is lighter than air, which is why it is used to fill helium balloons.Īrgon is used inside double-glazed windows as it doesn’t conduct heat. Lithium is a metal but is so soft it can be cut with a knife. Radium was once used to paint glow-in-the-dark hands on clocks and watches. Fun Facts about ElementsĪn element’s atomic number is the number of protons one atom of the element contains. Many of the elements at the bottom of the table ( in green ) are radioactive. Neon is used for neon signs, helium is used in balloons, argon is used in 3D printing, welding, lasers and has lots of other manufacturing uses. Noble gases are monatomic, they exist as single atoms. They are unreactive as they have a full outer shell of electrons and are very useful. One example of this is sodium reacting with chlorine to form sodium chloride. Halogens react with group 1 metals to create compounds. Fluorine is a yellow gas, bromine is a red liquid, and iodine is a purple solid. The halogen column is the only column to contain elements that are solids, liquids and gases. Halogens are the elements in light purple. These elements are very useful as they conduct electricity and are mostly solid at room temperature. The yellow block of elements in the centre of the table is known as transition metals. Pure sodium fizzes and shoots around when placed in water, potassium bursts into flame in water and caesium explodes when added to water! The reactivity of the elements increases as you go down the column. The elements in these groups are known as reactive metals. 
The red column is known as Group 1, and the orange column is Group 2. Lead has the symbol Pb – this comes from the Latin word plumbum. Tungsten has the symbol W – this is because tungsten is wolfram in German The first letter is always written as a capital, and the second is lowercase. Some are obvious, such as Li for Lithium, but some are harder to decipher. Chemical symbols that don’t look like their nameĮach element has a chemical symbol that is the same in every language. The modern periodic table is slightly different to the one created by Mendeleev as it is ordered by atomic number, not atomic mass. Mendeleev was so clever at spotting patterns he left gaps for undiscovered elements. The Periodic Table was invented by Dmitri Mendeleev in 1869, who was the first scientist to put elements in order of atomic mass. Metals are on the left of the table, metalloids are in the middle, and nonmetals are on the right.Įach square of the periodic table contains information about the element. Hydrogen is the first element in the Periodic Table, as its atom contains one proton. How are elements numbered in the Periodic Table?Įlements are numbered from left to right in order of how many protons their atoms contain. Elements with similar properties are grouped together. The periodic table currently contains 118 different elements. How many elements are there in the Periodic Table? 
Group 1 elements have 1 outer shell electron, group 2 has two outer shell electrons, and group 0 has full other shells with 8 electrons ( or 2 for Helium ). The group an element belongs to corresponds to the number of electrons in its outer shell. Groups have similar physical and chemical properties. The rows of the periodic table are called periods, and the columns are known as groups. 
The periodic table is a way of listing and organising elements. 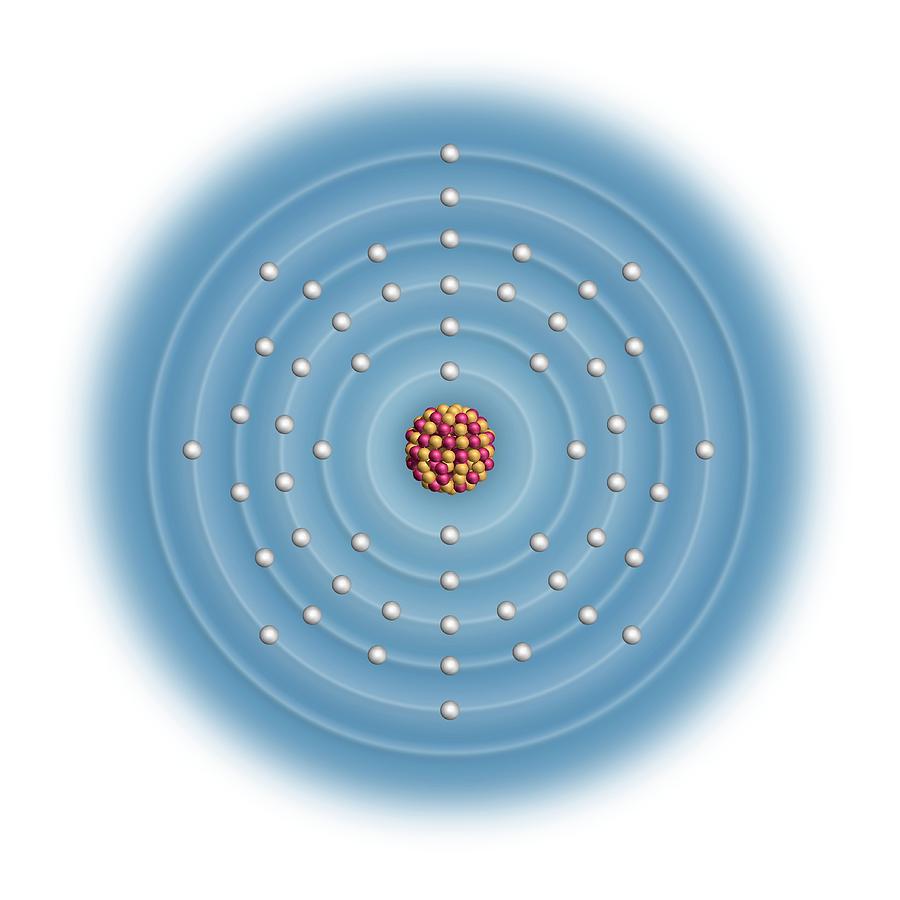
Elements are placed in order of atomic number ( number of protons in the nucleus ). The Periodic Table is a table of all the elements we know about today.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
